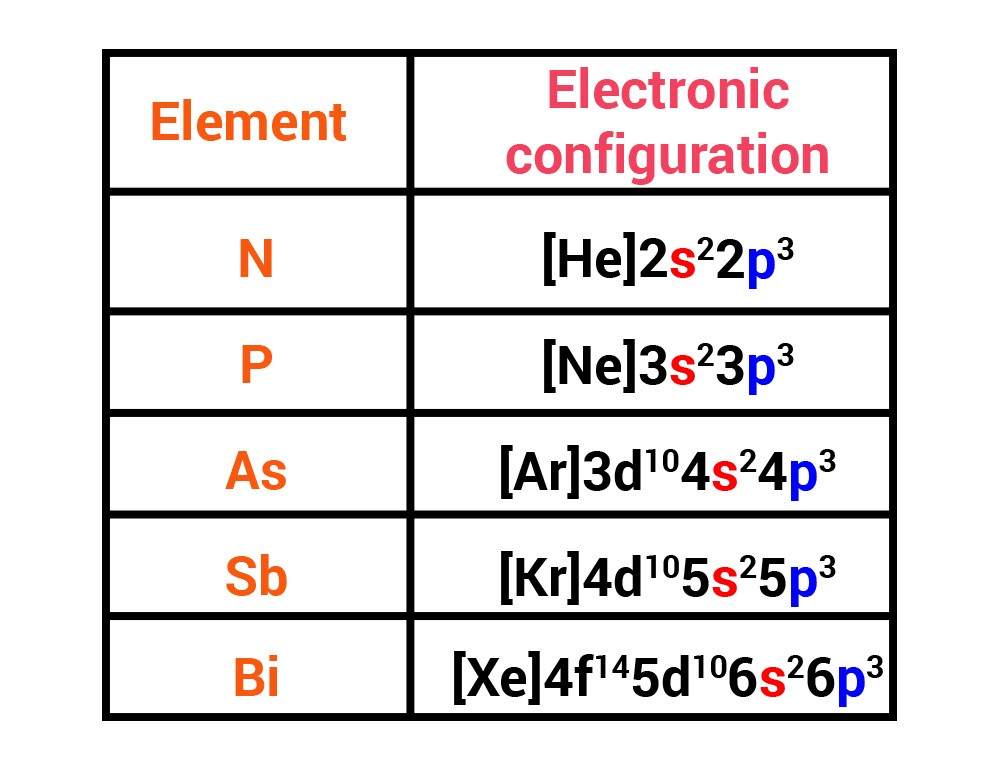

In a multi electron atom, the electrons present between the nucleus and the valence shell i.e inner electrons shield the valence electrons from the nucleus. This tendency to show higher valencies is less in Tl, Pb & Bi but more in Sn, In & Sb etc. When sufficient energy is available, the s-electrons also enter into bond formation and higher valencies are observed. The reluctance of s-electron pair to take part in bond formation is known as ‘inert pair effect’. In case of lower valencies, only electrons present in p-subshell are lost and s-electrons remain intact. What is the molar mass of the gas?Ĭalculate the temperature of 4.0 mol of a gas occupying 5 dm 3 at 3.32 bar.Some heavier elements of the III, IV & V group having configuration of outer most shell ns2 np1 ns2 np2 ns2 np3 show valencies with a difference of 2 i.e (1 & 3) ( 2 & 4 ) ( 3 & 5) respectively. What is meant by the term bond order? Calculate the bond order of: N 2, O 2,O 2 +,and O 2 -.Ģ.9 g of a gas at 95 ☌ occupied the same volume as 0.184 g of dihydrogen at 17 ☌, at the same pressure. Justify the position of hydrogen in the periodic table on the basis of its electronic configuration.įor an isolated system, ΔU = 0, what will be ΔS? (ii) Calculate the mass and charge of one mole of electrons. (i) Calculate the number of electrons which will together weigh one gram. How does atomic radius vary in a period and in a group? How do you explain the variation? Can CO 2, a well known fire extinguisher, be used in this case? Explain. Saline hydrides are known to react with water violently producing fire. Why is Wurtz reaction not preferred for the preparation of alkanes containing odd number of carbon atoms? Illustrate your answer by taking one example. Recently Viewed Questions of Class 11 ChemistryĪddition of HBr to propene yields 2-bromopropane, while in the presence of benzoyl peroxide, the same reaction yields 1-bromopropane. (ii) Determine the molality of chloroform in the water sample. The level of contamination was 15 ppm (by mass). What will be its density at STP?Ī sample of drinking water was found to be severely contaminated with chloroform, CHCl 3, supposed to be carcinogenic in nature. What mass of CaCO 3 is required to react completely with 25 mL of 0.75 M HCl?ĭensity of a gas is found to be 5.46 g/dm 3 at 27 ☌ at 2 bar pressure. Which one of the following will have largest number of atoms?Ĭalcium carbonate reacts with aqueous HCl to give CaCl 2 and CO 2 according to the reaction,ĬaCO 3(s) + 2 HCl (aq) → CaCl 2(aq) + CO 2(g) + H 2O (l) (iii) 100 atoms of A + 100 molecules of B In a reaction A + B2 → AB2 Identify the limiting reagent, if any, in the following reaction mixtures. (d) Cr 2O 7 2– + SO 2(g) → Cr 3+ (aq) + SO 4 2– (aq) (in acidic solution)ĭetermine the empirical formula of an oxide of iron which has 69.9% iron and 30.1% dioxygen by mass. (c) H 2O 2 (aq) + Fe 2+ (aq) → Fe 3+ (aq) + H 2O (l) (in acidic solution) (b) MnO 4 – (aq) + SO 2 (g) → Mn 2+ (aq) + HSO 4 – (aq) (in acidic solution) (a) MnO 4 – (aq) + I – (aq) → MnO 2 (s) + I 2(s) (in basic medium) (iii) 2 moles of carbon are burnt in 16 g of dioxygen.Ĭalculate the wavelength of an electron moving with a velocity of 2.05 × 10 7 ms –1.īalance the following redox reactions by ion – electron method : (ii) 1 mole of carbon is burnt in 16 g of dioxygen. is 3.0 × 10 –25 J, calculate its wavelength.Ĭalculate the amount of carbon dioxide that could be produced when The mass of an electron is 9.1 × 10 –31 kg.
Thus, its atomic number is 54 + 7 + 2 + 1 = 64. Its electronic configuration is 54 4 f 7 5 d 1 6 s 2. It belongs to group 3 of the periodic table since all f-block elements belong to group 3. It is an f –block element as the last electron occupies the f–orbital. (iii) Since n = 6, the element is present in the 6 th period. Hence, the element is Titanium ( 183d 24s 2. Therefore, it is a 4 th period and 4 th group element. = Number of s–block groups (4s 2) + number of d–block groups (3d 2) Thus, the corresponding group of the element It is a d–block element as d–orbitals are incompletely filled. (ii) Since n = 4, the element belongs to the 4 th period. Hence, the element is Sulphur ( 103s 23p 4) Therefore, the element belongs to the 3 rd period and 16 th group of the periodic table. = Number of s–block groups (3s 2) + number of d–block groups ( 10 + number of p–electrons(3p 4) There are four electrons in the p–orbital. It is a p–block element since the last electron occupies the p–orbital. (i) Since n = 3, the element belongs to the 3 rd period.
